Scalp cooling system for cancer patients gains regulatory approval in US
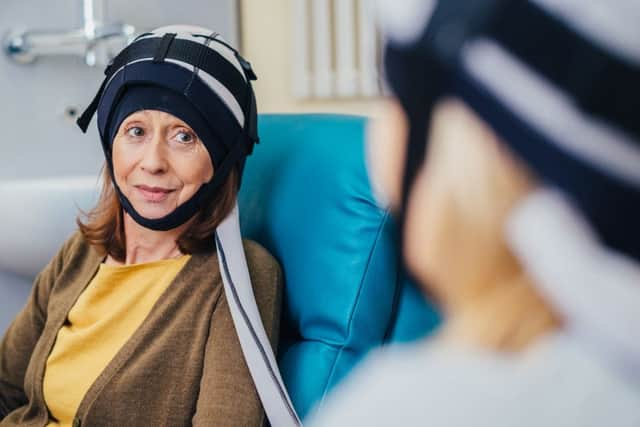

Paxman Coolers Limited has announced that The US Food and Drug Administration (FDA) has cleared the company to market the Paxman Scalp Cooling System, a technology that can reduce hair loss in breast cancer patients undergoing chemotherapy.
Paxman Coolers is a Huddersfield-based company which has established a worldwide reputation for its pioneering technology used by patients undergoing cancer treatment.
Advertisement
Hide AdAdvertisement
Hide AdAlthough established in the 1950s as Brewfitt, which still specialises in manufacturing cooling equipment for the brewing industry, the family-run business diversified in 1992 when Richard Paxman’s mother, Sue was diagnosed with breast cancer.
Intensive chemotherapy for aggressive breast cancer led to Mrs Paxman losing much of her hair, and prompted her husband Glenn and his brother Neil to use their technical expertise to develop a scalp cooler that reduces hair-loss in cancer patient
Mrs Paxman died in 2000 but in the intervening eight years Glenn and Neil had harnessed their inventive abilities to transform the unwieldy and largely ineffective cooling systems already in use to produce a cooling cap that now sets the industry standard.
Hair loss occurs because chemotherapy targets cancer tumours and all other rapidly dividing cells, including hair follicles. Cooling the hair follicles before, during and after treatment can dramatically reduce hair loss.
Advertisement
Hide AdAdvertisement
Hide Ad“Hair loss is consistently ranked in the top five most distressing cancer chemotherapy side effects,” said Richard Paxman, the CEO at Paxman, who is Sue’s son.
“Like my mum, many people find hair loss to be extremely traumatic. It is estimated that 8 per cent of patients actually refuse chemotherapy because they do not want to lose their hair.
“After experiencing this first hand, we have been determined to change this, and help minimise hair loss in women who are undergoing chemotherapy for breast cancer, positively contributing to their overall health and recovery.”
As part of the FDA clearance process, the Paxman scalp cooler was used in the first-ever randomised clinical trial to evaluate modern scalp cooling, which took place at a number of sites in the US. The multi-centre prospective study, which involved 186 women across New Jersey, New York, Texas, and Ohio, revealed that the cold cap preserved hair in more than 50 per cent of the women who used it.
Advertisement
Hide AdAdvertisement
Hide AdSpeaking about the results the lead researcher Dr Julie Nangia, MD, Assistant Professor of Medicine at Baylor College of Medicine, Houston, Texas, USA added: “The Paxman Hair Loss Prevention System is a safe and effective method for reducing hair loss in women being treated with chemotherapy for breast cancer, especially for those on taxane-based regimens.”
“Hopefully in five years from now, we will consider scalp cooling part of routine practice, the same way that we can see an IV-pump with an IV-pole as part of the regular equipment you would expect in an infusion suite. It’s important that people undergoing chemotherapy understand what scalp cooling is and that it is an option available to them if they want to prevent hair loss.”
Over the next 12 months, Paxman plans to install 250 systems across the US and will be working with a large number of cancer centres and large community oncology groups to roll out their scalp cooling systems.
A spokesman added: “The company aims to revolutionise the current landscape of scalp cooling by ensuring that affordability is at the centre of the treatment giving more patient choice.
Advertisement
Hide AdAdvertisement
Hide Ad“This involves launching a single patient use personal cap model, which will be loaded with scalp cooling credits depending on the number of chemotherapy infusions they are having. Patients using scalp cooling treatment will only pay for their own cap and the treatment they use.”
Richard Paxman added: “The US is the largest healthcare market in the world with over 1.6 million diagnoses of cancer each year. We have spent six years conducting a comprehensive multi-centre randomised clinical trial to ensure our data is as robust as possible. This has been a significant investment for us, but we are incredibly excited to be able to offer scalp cooling to US patients giving them a choice to maintain some control during treatment as we see in the UK and other parts of the world.”
Paxman Scalp Cooling technology, with more than 2,500 systems in hospitals, clinics, and treatment centres around the world, is made from lightweight, biocompatible silicone. The scalp cooling cap is soft and flexible, providing a snug yet comfortable fit during treatment.