Yorkshire mother says her son slept through 'world's most expensive treatment' and is 'doing ok'
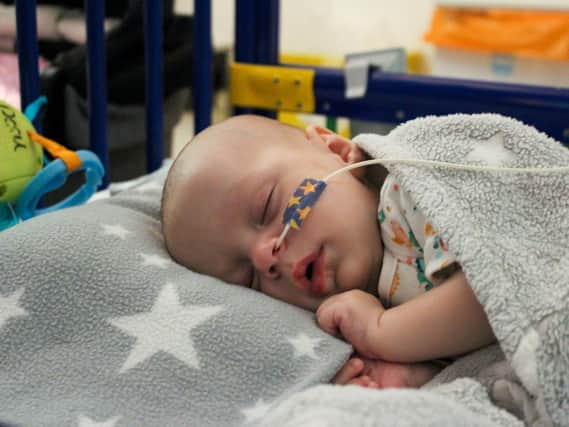

Mother Gunita said baby Markuss, who has Type 1 SMA, slept through the one-off, hour-long gene-therapy treatment with the potentially life-saving drug Zolgensma at Sheffield Children’s Hospital.
She thanked everyone at the hospital – which is one of four centres in England approved for the use of the drug for the treatment of Type 1 SMA – for saving her son’s life.
Advertisement
Hide AdAdvertisement
Hide AdGunita, who is from York, said: “The health visitor noticed that something was wrong soon after he was born. Markuss was tested at our local hospital, where they told me that he had Type 1 SMA – that was when he had just turned two months old.
“We were then told about this treatment and decided to go for it. Two weeks after that, we were in the hospital having the gene-therapy treatment. Everything happened so fast and quick.
“The whole infusion thing was over in an hour and Markuss slept through all of it. I was really nervous before but he’s doing OK. It’s just the one treatment – they gave the missing gene back to him.
“Everyone at Sheffield Children’s Hospital has been just awesome. I’d like to say thank you to everyone for saving his life.”
Advertisement
Hide AdAdvertisement
Hide AdA hospital spokeswoman said Zolgensma, which has a list price of £1.79 million per dose and is known as the “world’s most expensive drug”, can provide improvement in motor function and prolong the lives of children with a specific type of SMA – a rare and often fatal genetic disease that causes paralysis, muscle weakness and progressive loss of movement.
Markuss received the drug through a drip inserted into a vein and should be able to go home around two weeks after receiving the treatment following post-infusion check-ups.
The spokeswoman said the expectation is that Markuss will now have a better chance of sitting, crawling and walking, things that children with Type 1 SMA struggle to do.
NHS England said eight babies had received the treatment since it struck a landmark deal with manufacturers Novartis Gene Therapy in March to make Zolgensma available on the NHS.
Advertisement
Hide AdAdvertisement
Hide AdThe four Trusts commissioned to administer the treatment – in Sheffield, London, Manchester and Bristol – will work together in collaboration with regional neuromuscular centres across the country to share expertise and experience with the new therapy.
Jeff Perring, medical director at Sheffield Children’s Hospital, said: “We are delighted to be able to offer this treatment to young patients affected by spinal muscular atrophy.
“This treatment will really improve the quality of life for these children and we’re proud to be part of this great advance in treatment using gene therapy.”