Here's how the NHS Covid-19 antibody test works and which shops are selling their own test kits

Health Secretary Matt Hancock announced on 21 May that the Government had agreed a deal with pharmaceutical firms Roche and Abbott for more than 10 million antibody tests.
Public Health England (PHE) said the test has an accuracy of 100 per cent, with the findings hailed as a “very positive development” in tackling the virus outbreak.
Advertisement
Hide AdAdvertisement
Hide AdUntil now health officials have said such tests are not reliable enough, but after evaluation of the Roche test last week, PHE found it to be “highly specific”.
The blood test has been developed by Swiss pharmaceutical company Roche and is designed to determine if a person has been exposed to the virus.
Here's everything you need to know about it:
How does the test work?
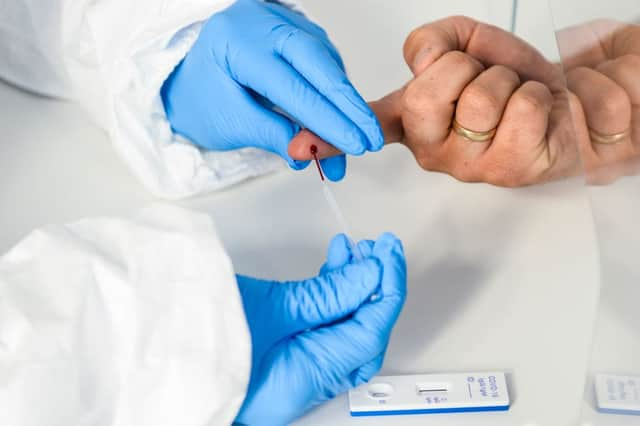
The test involves taking a small sample of blood and testing it for antibodies which will indicate exposure to coronavirus.
The test picks up 100 per cent of people who have had coronavirus, which means it has 100% sensitivity.
Advertisement
Hide AdAdvertisement
Hide AdIt also has a specificity of over 99.8 per cent, meaning it picks up virtually all people who have not had coronavirus; a test that is 100 per cent specific means all healthy individuals are correctly identified as healthy - there are no false positives.
As it is able to detect 100 per cent of cases where a person has had coronavirus in the past, the test could prove useful in helping to inform when lockdown restrictions may be safe to ease.
When will the test be available?
The new antibody test is expected to first be available in the UK over the next few weeks.
The Government is “confident” of being able to handle thousands of the antibody tests every day from 1 June. However, the full roll out could take a number of months.
Advertisement
Hide AdAdvertisement
Hide AdRoche said it could supply hundreds of thousands of the tests each week, with the company first prioritising tests for distribution via the NHS, before looking at how they may be sold to individuals.
Who can get the test?
Health minister Edward Argard said the new test is intended to be rolled out to frontline NHS workers first; the government plans to make the test available to workers in social care after the NHS and then distribute it more widely.
Although it is unclear how and when it will happen, it is hoped the test will eventually become available to the wider public; for now, people who are having blood tests for other ailments will be asked if they would also like a coronavirus antibody test.
What about Superdrug’s test?


Superdrug recently introduced its own antibody test, though concerns have been raised over the accuracy of results with the chain saying it will detect Covid-19 antibodies in 97.5 per cent of cases.
Advertisement
Hide AdAdvertisement
Hide AdA blood sampling kit is sent in the post, in which there are detailed instructions about how to carry out the test with a simple finger prick.
A few drops of blood need to be collected into a small vial, secured in the packaging, and posted to the UKAS-accredited lab using the prepaid return packaging.
But Abbott, the manufacturer of the test, said it was not intended to be used in this way, and the test should only be administered by a medical professional.
Anyone who is over the age of 18 can take the test, and the Superdrug Covid-19 Antibody Blood Test is priced at £69 and is available through Superdrug's Online Doctor service.
Are you immune from Covid-19 if you have the antibodies?
Advertisement
Hide AdAdvertisement
Hide AdIt remains unclear what level of immunity people develop once they have had Covid-19; scientists are unsure and there is still a lot to learn about coronavirus.
Experts believe that while the presence of antibodies indicates a level of immunity, it is unclear whether people are completely protected and how long any immunity lasts.
There has been some suggestion that immunity could last for two to three years but more research needs to be done.
Will the test be available outside England?
Health officials in Scotland, Wales and Northern Ireland make their own decisions about testing, but are likely to roll out the antibody test if England adopts it.
Advertisement
Hide AdAdvertisement
Hide AdThe Government says it will arrange supplies of tests on behalf of the devolved nations, who can decide how to use their allocations.
The test also already has approval from medical regulators in the EU and the United States.